
Two decades from Yamanaka's lab to the world's first commercial approval. Sources: Kyoto University CiRA, Nobel Committee, Japan MHLW
On March 6, 2026, exactly 20 years after the first mouse induced pluripotent stem cells were created in a Kyoto University laboratory, Japan's Ministry of Health, Labor and Welfare approved two medical products derived from those same reprogrammed cells. The timing was not accidental. It was the capstone of a national scientific project that stretched across two decades, one Nobel Prize, and hundreds of millions of dollars in public and private investment.
The two approved products - ReHeart, a sheet of cardiac muscle cells for severe heart failure, and Amusepri, dopamine-producing neurons for Parkinson's disease - are the first commercial applications of iPS cell technology anywhere in the world. They will not be available in pharmacies tomorrow. Insurance coverage negotiations will take months. Clinical data requirements remain strict. But the door has opened. And what comes through it will reshape medicine in ways that rival antibiotics or organ transplantation.
This is not a story about a promising laboratory experiment. This is a story about what happens when a technology crosses from theoretical possibility into something a doctor can prescribe.
What iPS Cells Actually Are - And Why They Matter So Much
The term "induced pluripotent stem cells" is a mouthful that obscures a remarkably simple and radical idea. Take an ordinary adult cell - a skin cell, a blood cell, a liver cell - and chemically reprogram it to behave like an embryonic stem cell. Suddenly you have a cell that can become anything. Heart muscle. Brain neurons. Liver tissue. Pancreatic beta cells. Bone. Cartilage.
Embryonic stem cells could already do this, and researchers had been working with them since the 1990s. But embryonic stem cells come from human embryos, which made their use a political and ethical minefield that blocked research, restricted funding, and created legislative obstacles in dozens of countries.
Shinya Yamanaka solved that problem in 2006. Working at Kyoto University, he discovered that just four transcription factors - now known as the Yamanaka factors - could reset the developmental clock of an adult cell, pushing it back to a pluripotent state without involving any embryo. In 2007, his team did the same with human cells. The scientific world erupted. In 2012, Yamanaka shared the Nobel Prize in Physiology or Medicine with John Gurdon, who had pioneered nuclear reprogramming decades earlier.
"We are very pleased to have taken a major step toward social application on the 20th anniversary of the announcement of mouse iPS cells. To establish it as a medical treatment, it is essential to go through the process of confirming its safety and efficacy in many more cases." - Shinya Yamanaka, Director Emeritus, iPS Cell Research Institute, Kyoto University
The promise was enormous. iPS cells could potentially allow doctors to grow patient-specific replacement tissue without rejection risk. They could model individual patients' diseases in a dish for drug testing. They could generate cell types that are otherwise impossible to obtain. But turning laboratory promise into clinical reality took two more decades of hard work, regulatory navigation, and infrastructure building that the Nobel citation could not rush.
ReHeart: A Cardiac Patch That Whispers to a Failing Heart
The first of the two approved products is ReHeart, developed by Qualipse, a startup founded at Osaka University. The product consists of sheets of cardiomyocytes - specialized heart muscle cells - differentiated from iPS cells obtained from healthy donors.
The target population is severe heart failure patients whose condition stems from ischemic cardiomyopathy - heart damage caused by blocked blood supply - and who have not responded adequately to standard treatments including drug therapy and invasive procedures. For these patients, the realistic options before ReHeart were a heart transplant, which is severely limited by donor shortages and age restrictions, or a mechanical heart assist device, which carries persistent risks of infection, stroke, and long-term quality-of-life decline.
ReHeart takes a different approach. Three sheets of cardiomyocytes are attached to the surface of the heart during a surgery performed through the left side of the chest. The transplanted cells do not simply replace dead tissue mechanically. They secrete signaling proteins - paracrine factors - that stimulate blood flow, reduce inflammation, and help recruit the heart's own repair mechanisms.
In the national multicenter collaborative study that formed the basis for approval, eight patients with severe heart failure received the treatment. Four showed measurable improvement, with peak oxygen consumption - a key indicator of cardiac function - increasing by more than 10 percent at 52 weeks post-transplant. (Source: Journal of the Japanese Medical Association, 2025)
ReHeart - Key Facts
- Developer: Qualipse (Osaka University spinout)
- Cell type: iPS-derived cardiomyocytes from healthy donors
- Delivery: Three cell sheets sutured to heart surface via left chest incision
- Trial results: 4 of 8 patients improved; VO2 peak +10% at 52 weeks
- Commercial study required: 75 patients, outcome tracking
- Insurance coverage target: Fall 2026
- Approval type: Conditional and time-limited (Japanese regenerative medicine track)
The approval is conditional and time-limited, meaning Qualipse must run a post-marketing study targeting 75 patients and report outcomes before full permanent authorization is granted. Sales are expected to begin around autumn 2026, once insurance coverage is negotiated. The product generated significant public attention when it was exhibited at the Osaka-Kansai Expo earlier this year.
The second-order effect here is not the 75 patients in the initial commercial study. It is what happens when the data from those patients accumulates and enables broader coverage approvals, when manufacturing scales, and when the basic concept - using lab-grown cell sheets to repair damaged organs - gets applied to the liver, the kidney, the retina. ReHeart is version one. The platform is what matters.
Amusepri: Seeding a Parkinson's Brain With New Neurons
Amusepri, developed by Sumitomo Pharma and Racthera, addresses a problem that has frustrated neurologists for decades: Parkinson's disease destroys dopamine-producing neurons in the substantia nigra, a region deep in the midbrain, and current drugs can only manage symptoms. They cannot replace what is lost.
Amusepri attempts to do exactly that. The product consists of dopamine neuron precursor cells derived from donor iPS cells. These precursors are injected directly into specific brain regions through a minimally invasive neurosurgical procedure in which small holes are drilled in the skull and the cells are delivered via three injection routes on each side of the brain.
The goal is that these precursor cells will mature into functional dopaminergic neurons, integrate into existing neural circuits, and begin producing the dopamine that the disease has depleted. This is a fundamentally different therapeutic logic than any drug: instead of compensating for missing dopamine chemically, Amusepri tries to restore the factory that was shut down.
"The results of the deliberations are a major step forward, but even if approval is granted, that is not the goal, but only the beginning of this new medicine." - Jun Takahashi, Director, iPS Cell Research Institute, Kyoto University
In the physician-led trial conducted at Kyoto University Hospital, six patients with Parkinson's disease were treated. Four of the six showed improvement in their off-time scores on the MDS-UPDRS Part III motor assessment - the gold-standard measure of Parkinson's motor symptoms - at 24 months post-transplantation. Crucially, cells were confirmed to remain viable in all six patients at the transplant site. (Source: Kyoto University CiRA press release, April 2025)
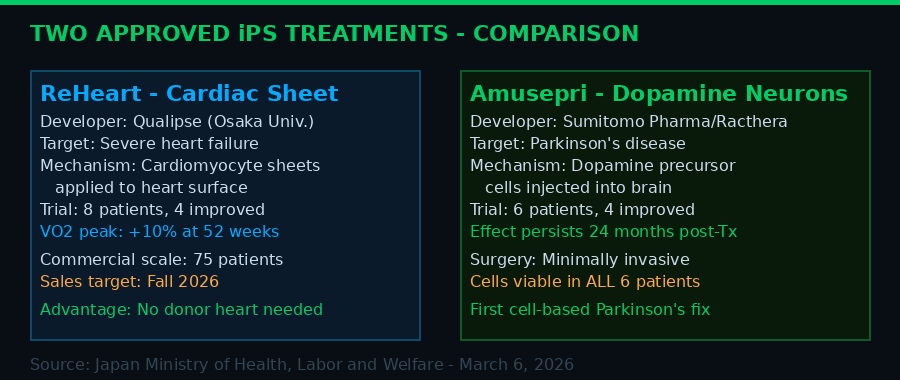
Side-by-side comparison of the two approved therapies. Both use donor iPS cells processed through Japan's first dedicated commercial manufacturing facility, SMaRT. Sources: Japan MHLW, Kyoto University CiRA, Sumitomo Pharma
The viability finding matters enormously. One of the longstanding fears about cell transplantation into the brain was immune rejection - the brain's microglia attacking foreign cells even in an immune-privileged environment. The fact that all six patients showed surviving cells at two years suggests the donor iPS cell approach, combined with immunosuppression protocols, is managing this risk. It is a proof-of-concept that the immune problem, while not trivially solved, is not a dead end.
The Infrastructure Behind the Milestone
What makes Japan's achievement notable is not just the science. It is the fact that an entire industrial and regulatory ecosystem was constructed to make it possible - and that this ecosystem was almost entirely domestic.
At the center of the supply chain is SMaRT, the world's first commercial manufacturing facility dedicated exclusively to regenerative medicine and cell-based drugs derived from donor iPS cells. Located in Suita City, Osaka Prefecture, SMaRT is responsible for producing Amusepri. The raw material - validated, quality-controlled iPS cells - comes from a stock maintained by the Kyoto University iPS Cell Research Foundation, which Yamanaka established specifically to create a GMP-grade cell bank for medical use.
The manufacturing process incorporates differentiation technologies developed at Kyoto University, along with cell purification technology licensed from Eisai, one of Japan's major pharmaceutical companies. This web of industry-academia collaboration - from basic research to cell banking to differentiation protocols to quality control to clinical execution - was assembled over more than a decade. It did not happen by accident.
Japan's iPS Cell Supply Chain - End to End
- Basic technology: Yamanaka lab, Kyoto University (2006-present)
- Cell banking: Kyoto University iPS Cell Research Foundation (GMP-grade donor iPS stock)
- Manufacturing: SMaRT facility, Suita City - first dedicated commercial facility in the world
- Differentiation tech: Kyoto University proprietary protocols
- Cell purification: Eisai technology integrated into Amusepri process
- Clinical development: Osaka University (ReHeart), Kyoto University Hospital (Amusepri)
- Commercial partners: Qualipse (Osaka Univ. spinout), Sumitomo Pharma, Racthera
- Regulatory pathway: Japan's conditional approval system for regenerative medicine
Japan's regulatory framework deserves specific attention here. In 2014, Japan introduced an accelerated approval pathway for regenerative medicine products - the "conditional and time-limited" authorization system visible in both approvals. This system allows products with strong preliminary safety and efficacy signals to enter limited commercial use while post-marketing studies are conducted. It is a deliberate trade-off: patients get access faster, and real-world data accumulates faster, in exchange for ongoing reporting requirements and the possibility of revocation if the evidence does not hold up.
Critics of the system argue it risks exposing patients to therapies that have not yet proven themselves at scale. Proponents point out that waiting for traditional large-scale randomized controlled trial data - standard in drug development - is impractical and ethically questionable when the patient population is severely ill, the alternatives are exhausted, and the safety profile looks acceptable. Japan has been running this experiment for more than a decade. The two March 2026 approvals are its most significant output yet.
Why the Ethical Bottleneck No Longer Exists
The approval of iPS-derived therapies represents something that goes beyond a single medical milestone. It resolves, at least in part, the central ethical problem that had paralyzed stem cell research for a decade.
In the early 2000s, the potential of embryonic stem cells was scientifically clear and politically explosive. The Catholic Church, the evangelical right, and many secular bioethicists objected to research that required destroying human embryos. The United States, under the Bush administration, restricted federal funding for embryonic stem cell lines created after August 2001. Some European countries banned the research entirely. Labs that wanted to work with embryonic stem cells faced a labyrinth of regulations, funding restrictions, and in some places criminal liability.
Yamanaka's 2006 discovery changed the calculus. iPS cells offer essentially the same pluripotent capabilities as embryonic stem cells - the ability to become any cell type in the body - without requiring an embryo at any stage of the process. A skin cell is reprogrammed. No embryo is created. No embryo is destroyed. The ethical objection that blocked a decade of research simply does not apply.
This matters beyond Japan. The approval signals to researchers, regulators, and investors worldwide that the iPS technology pathway is clinically viable. Expect regulatory agencies in the United States, the European Union, South Korea, and elsewhere to accelerate their own review frameworks. The FDA has been watching Japan's conditional approval experiment closely. The March 2026 decision gives American regulators real-world outcome data they did not have before - and a political cover to move forward on therapies that would have faced fierce opposition if derived from embryonic cells.
The Diseases That Come Next
ReHeart and Amusepri are the first approvals. They will not be the last. The iPS cell research pipeline in Japan and internationally includes treatments targeting conditions that currently offer patients few or no options.
Age-related macular degeneration was actually the first condition where iPS cells were used in human clinical trials, back in 2014 at RIKEN in Japan. Photoreceptor replacement using iPS-derived retinal pigment epithelium cells has been in trials at multiple institutions. Spinal cord injury is another active area - iPS-derived neural precursors have been transplanted into patients with sub-acute complete spinal cord injuries in a Keio University trial. Type 1 diabetes is being targeted with iPS-derived pancreatic beta cells. Even sickle cell disease and other blood disorders could be addressed through patient-specific iPS cells that are gene-corrected before differentiation.
The Parkinson's approval is particularly significant because it establishes proof of concept for neurological applications - arguably the hardest category. If you can successfully transplant viable cells into the brain and have them function two years later, you have demonstrated that the brain-transplantation technical barrier is not insurmountable. That finding unlocks the theoretical pathway for treating Huntington's, ALS, spinal muscular atrophy, and a range of other neurodegenerative conditions through cell replacement approaches.
"Twenty years have passed since the emergence of iPS cells. The technology, which originated in the laboratory, has reached the stage where human cells are cultured, differentiated, and finally administered to the patient's body." - Wired Japan, March 14, 2026
The economic implications run alongside the medical ones. The global regenerative medicine market was valued at roughly $25 billion in 2023 and was growing at 15-20 percent annually even before the Japan approvals. Manufacturing scale-up is the primary bottleneck - producing clinical-grade cell products at volume is expensive and technically demanding. But the approval of commercial products creates financial incentive for capital investment in manufacturing infrastructure that research programs alone could never attract.
What Still Has to Go Right
It would be a mistake to read the March 6 approvals as a solved problem. They are a beginning, and a conditional one.
Both ReHeart and Amusepri were approved under Japan's conditional framework, which means the evidence base is small - eight patients for one, six for the other. The improvement signals are real and encouraging, but the sample sizes that formed the approval basis would not pass muster in a standard Phase III drug approval process in the United States or the European Union. The post-marketing studies that Japan requires are not optional extras; they are fundamental to establishing whether these treatments actually work at scale.
The immune rejection question is managed but not fully resolved. Both products use donor iPS cells, not patient-specific cells. Donor cells avoid the logistical nightmare of manufacturing a custom product for each individual patient - a process that takes months and costs hundreds of thousands of dollars. But donor cells, even from carefully matched sources, require immunosuppression, which carries its own risks including infection susceptibility and long-term organ stress. Whether the matched donor approach is good enough, or whether certain patients will still reject the cells despite immunosuppression, will only become clear as patient numbers accumulate.
Manufacturing reproducibility is another challenge. Differentiating iPS cells into specific cell types requires exquisite process control. A cardiac muscle cell sheet that deviates from protocol specifications is not the same as a batch of tablets where the active ingredient is off by a few percent. The SMaRT facility in Osaka is the world's first dedicated commercial plant for these products. There is no established playbook for running it at scale, no decades of accumulated manufacturing knowledge. The first hundred patients will generate as much manufacturing data as medical outcome data.
And then there is access. These will not be cheap. Regenerative medicine products at this level of biological complexity and manufacturing difficulty will almost certainly cost more than conventional drugs, possibly dramatically more. Japan's universal health insurance system provides a potential mechanism for broad access once coverage is established - the expected three-to-five month timeline for insurance listing after approval. But the global picture is different. In countries without universal coverage, iPS cell therapies could easily become available only to the wealthy, replicating the access inequities already seen with CAR-T cancer therapies that cost $400,000 to $600,000 per treatment in the United States.
The Moment the Era Begins
In the history of medicine, there are a handful of moments that divide what came before from what came after. The discovery of penicillin in 1928. The Salk polio vaccine in 1955. The first successful kidney transplant in 1954. The introduction of statins for cardiovascular disease. Each of these did not eliminate the disease it targeted overnight. Each required years of refinement, infrastructure building, manufacturing scale-up, and regulatory evolution before it became standard care. Each is now unremarkable - routine - in a way that seemed impossible when it began.
The Japan approvals of March 6, 2026 may belong in that list. Not because ReHeart and Amusepri will immediately cure heart failure and Parkinson's at scale. They will not - not yet. But because they establish something that no regulatory body anywhere in the world has established before: that a human cell can be taken, reprogrammed back to an embryonic-like state, guided to become a specialized cell type, manufactured at commercial scale, transplanted into a human body, and help that body function better than it did before.
That is a new category of medicine. Japan, where the Nobel Prize winner did his work, where the cell banks were built, where the regulatory framework was specifically designed to enable this moment, got there first.
Kenichiro Ueno, Japan's minister of health, labor, and welfare, said it plainly at his March 6 press conference: "I am very pleased that a therapeutic product from Japan based on professor Yamanaka's iPS cells has been put into practice for the first time in the world, and I hope it will help patients not only in Japan but all over the world."
Jun Takahashi, who led the Amusepri trial at Kyoto University, added a note of scientific caution that researchers everywhere should take seriously: "Even if approval is granted, that is not the goal, but only the beginning of this new medicine."
He is right. The beginning has arrived. What medicine looks like at the end of this era, nobody can say yet. But the cells are already in the body. The clock is running.
Get BLACKWIRE reports first.
Breaking news, investigations, and analysis - straight to your phone.
Join @blackwirenews on Telegram