India's Ozempic Moment: The Patent That Will Change Everything
On March 20, semaglutide's Indian patent expires. Fifty generics are ready to flood the market. For 180 million overweight Indians, the math just changed. But cheaper pills don't cure a culture that glorified the pot belly for centuries - and the people most likely to get hurt aren't the ones you'd expect.

BLACKWIRE illustration. India stands two days away from a pharmaceutical inflection point that analysts are calling the biggest health market shift in a generation.
The 23-year-old man who walked into Dr. Rahul Baxi's Mumbai clinic last November didn't come in sick. He came in scared. He'd gained 10 kilograms since starting a corporate job. His gym buddy was already on the jabs. He wanted in.
Dr. Baxi said no. He told the young man what would happen when he stopped: appetite returns hard, the weight comes back with interest, and without proper exercise he'd lose muscle alongside fat. The man left disappointed. But he is the story of what's about to happen at scale.
This Thursday, March 20, semaglutide - the molecule behind Ozempic and Wegovy, the most talked-about weight-loss drugs in the world - loses its patent protection in India. Within months, around 50 branded generics are expected to flood the market. Prices are set to drop by more than half. And the country with the world's second-highest number of overweight adults, 180 million people, will suddenly be able to afford a drug that was previously the privilege of the upper-middle class.
Investment bank Jefferies is calling it a "magic pill moment" for India, projecting the semaglutide market could reach $1 billion domestically within a few years. (Source: Jefferies analyst note via BBC, March 2026.) The word "moment" feels right. But moments cut both ways.

Key data points behind India's weight-loss crisis, sourced from Pharmarack, Lancet, NFHS-5 national survey, and Jefferies research.
The Body That Became a Warning Sign
To understand what's about to happen, you have to understand what the pot belly meant in India for most of living memory.
In literature, film, and rural folklore, a rounded stomach signalled prosperity. It meant this man eats well. Politicians' bellies were exaggerated in cartoons not as a health warning but as a mark of their comfortable corruption. The fat industrialist, the indulgent uncle, the well-fed landlord - these were culturally coded as figures of comfort, even aspiration.
That cultural coding is now colliding with hard medical data. India had the second-highest number of overweight or obese adults in 2021: 180 million. A 2025 Lancet study projected that number could reach 450 million by 2050 - nearly a third of the country's projected population. (Source: The Lancet, 2025.)
The shape of the problem is specific to South Asian bodies in ways that make it more dangerous than BMI charts reveal. Fat in Indian bodies tends to accumulate around the abdomen and, critically, around the organs. The liver, pancreas, and heart bear a disproportionate fat load. Studies show South Asians have more body fat than white Caucasians at the same BMI - and that fat is distributed in the worst possible places.
"As shocking as it may sound, even people with a normal weight can have dangerous levels of belly fat." - Dr. Anoop Misra, Director, Fortis-C-DOC Centre of Excellence for Diabetes, Metabolic Diseases and Endocrinology, Delhi
The numbers on abdominal obesity are already alarming. According to the National Family Health Survey (NFHS-5) - which for the first time measured waist and hip sizes - 40% of women and 12% of men in India have abdominal obesity. Among women aged 30 to 49, nearly one in two are already showing signs. (Source: NFHS-5, published in peer-reviewed analysis.)
One evolutionary theory, speculative but cited by doctors, holds that centuries of famine and food scarcity trained Indian bodies to store fat aggressively in the abdomen - an adaptation that now fires as a liability in an era of ultra-processed food. Between 2009 and 2019, India ranked among the world's fastest-growing markets for ultra-processed food sales, alongside Cameroon and Vietnam. The famine-adapted body and the Swiggy delivery app are in direct conflict.
A Drug That Changed the Game
Semaglutide belongs to a class of medicines called GLP-1 receptor agonists. The mechanism is elegant: they mimic a natural gut hormone that slows digestion and signals to the brain that the body is full. Users eat less, feel fuller faster, and the effect is sustained. Originally developed for Type 2 diabetes, these drugs were repurposed for obesity as trial after trial showed dramatic weight-loss results - typically 10-15% of body weight.
In India, Novo Nordisk's oral version Rybelsus launched in 2022 and almost immediately dominated the market. The injectable Wegovy followed. A monthly course of Wegovy currently costs between 10,000-16,000 rupees - roughly $108 to $173. Not impossible money for upper-middle-class urban professionals, but out of reach for most of India. Rybelsus accounts for nearly two-thirds of India's weight-loss drug market since its launch. (Source: Pharmarack research firm, cited by BBC, November 2025.)
The market went from $16 million in 2021 to nearly $100 million by 2025 - a sixfold surge in four years. That was with prices high and supply limited. Now comes the flood.
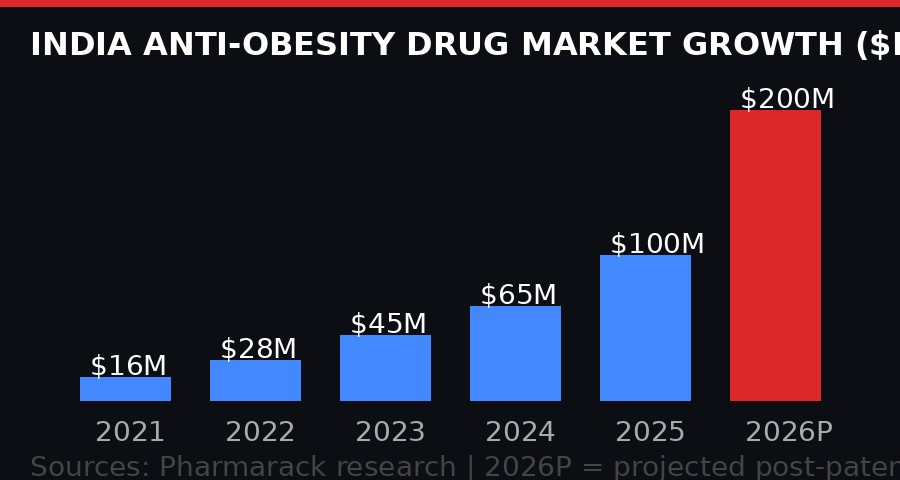
India's anti-obesity drug market grew sixfold in four years at premium prices. The 2026 projection reflects analyst expectations following generic market entry. Source: Pharmarack.
Fifty Generics and the Race to the Bottom
When sitagliptin - a diabetes drug - went off patent in India in 2022, roughly 30 branded versions appeared within one month and nearly 100 within a year. India's generic pharmaceutical machine runs at a pace no other country matches. Sheetal Sapale, vice-president of research at Pharmarack, says major firms including Cipla, Sun Pharma, Dr Reddy's Laboratories, Biocon, Natco, Zydus and Mankind Pharma are already preparing branded generics. Many more smaller players are expected to follow.
Price forecasts: generic competition is expected to push the monthly treatment cost from the current 8,800-16,000 rupees range down to roughly 3,000-5,000 rupees ($36-54) per month. (Source: Sheetal Sapale, Pharmarack, via BBC March 2026.)
That is a meaningful shift. Not cheap by global standards of health access, but a significant reduction that puts the drug within reach of India's vast lower-middle class. Beyond India, the implications compound: India is the world's largest supplier of generic medicines, producing around 60,000 drug brands and accounting for roughly 20% of global generic supply. The country supplies medicine to over 200 nations, meeting more than half of Africa's generic drug demand and about 40% of generics used in the United States.
"The export potential of Indian generic weight-loss drugs is humongous. The US market alone could scale to $10 billion within a few years as obesity rates drive demand." - Namit Joshi, Chairman, Pharmaceuticals Export Promotion Council of India, via BBC
The HIV parallel is frequently cited. Two decades ago, Indian generic manufacturers slashed antiretroviral drug prices, dramatically expanding HIV treatment across Africa and the developing world. Many public health advocates are quietly hoping the same playbook works here. But HIV drugs didn't come with a social-media-fuelled demand problem, bridal slimming packages, or gym trainers operating as unlicensed pharmacists.
What's Coming In
50+ branded semaglutide generics expected within months
Major manufacturers: Cipla, Sun Pharma, Dr Reddy's, Biocon, Natco, Zydus, Mankind Pharma
Price target: Rs. 3,000-5,000/month (from Rs. 8,800-16,000)
Market could reach $1 billion domestically within 3-5 years
The Warning Signs
Gym trainers already prescribing and injecting GLP-1 drugs illegally
Beauty clinics offering "bridal slimming packages" - rapid weight loss before weddings
Online pharmacies dispensing after "cursory phone consultations"
Fears of counterfeit drugs entering the supply chain
Who Gets Hurt When the Price Drops
The wedding industry spotted the opportunity first. In urban India, there is already a thriving market for what clinicians politely call "event-driven weight loss." Beauticians and unqualified dietary consultants have begun advertising rapid slimming packages targeted at brides ahead of their wedding dates - packages that sometimes involve GLP-1 injections sourced through informal networks.
Dr. Bhaumik Kamdar, a Mumbai-based chest physician, recalls a father walking into his clinic asking whether the new drugs could help his daughter lose seven kilograms before her wedding - in three months. "He wanted to know if they really work," Kamdar says. The concern wasn't whether it was medically appropriate. The concern was whether it was effective enough, fast enough.
This is the social texture of what's coming. A drug designed and trialled for people with clinical obesity and Type 2 diabetes is rapidly becoming a lifestyle product, subject to the same pressures that drive crash dieting, cosmetic surgery, and fitness supplement abuse. The difference is that semaglutide works in ways those things mostly don't - which makes unsupervised misuse more consequential.
The side effects are real and documented. Nausea, vomiting, and digestive disruption are common, particularly at higher doses. Rarer complications include gallstones and pancreatitis. Most seriously, rapid weight loss without adequate protein intake and exercise causes muscle loss alongside fat loss - a particular concern in India given what doctors call "sarcopenic obesity," the simultaneous presence of low muscle mass and high body fat.
"More access to cheap drugs means a higher chance of abuse. Access calls for more responsibility - and stricter regulation. I am cautiously optimistic about these drugs." - Dr. Bhaumik Kamdar, chest physician, Mumbai, via BBC
And then there is the rebound problem. When patients stop semaglutide, appetite returns aggressively. The body fights hard to restore lost weight. "If you stop the drugs, the appetite comes back voraciously," Dr. Baxi says. Without the lifestyle changes - the protein, the exercise, the habits - patients regain the weight. Then some try to go back on the drug. The cycle begins.
Studies show these drugs are most effective as long-term treatments, not short courses. But long-term pharmaceutical dependence at $36-54 a month, across hundreds of millions of potential users, raises its own questions about sustainability and access equity. Who stays on it? Who can afford to stay on it? What happens to the person who starts, loses 12 kilograms, then loses their job and can no longer afford the monthly injection?
The Regulation Gap India Cannot Afford
India's drug regulator moved last week. An advisory warned pharmaceutical companies against promoting prescription weight-loss medicines directly to consumers. Advertising that promises dramatic results or downplays the need for diet and exercise "could be deemed misleading," officials said, stressing these medicines should only be used under medical supervision. (Source: India Drugs Controller, advisory issued March 2026 via BBC.)
That advisory hits against a healthcare infrastructure reality that the language of regulation cannot paperover. India has approximately 1.2 doctors per 1,000 people - well below the WHO recommended ratio of 4.45. Rural areas are dramatically underserved. For the hundreds of millions of newly eligible patients outside urban centres, "medical supervision" means a brief video call, or a WhatsApp message to a local pharmacist, or nothing at all.
The specific worry is manufacturing quality. Dr. Muffazal Lakdawala, a Mumbai-based bariatric surgeon who has been at the centre of India's weight-loss drug conversation, puts it plainly: "The quality of the drugs being made here must be very tightly regulated. These are very beneficial drugs. We don't want side effects arising from poor-quality medicines and giving the molecule itself a bad name."
India's generic pharmaceutical industry has faced regulatory scrutiny before. The US Food and Drug Administration has flagged Indian manufacturing facilities on repeated occasions over the past decade - for data integrity issues, sterility violations, and contamination. In the semaglutide generics rush, the pressure to move fast, launch early, and capture market share may collide with the meticulous quality standards that injectable biologics demand.
The distinction between a legitimate generic Wegovy and a low-quality counterfeit is invisible to the patient receiving a weekly injection from their gym trainer. Side effects from poor-quality products could be severe. They could also discredit the entire drug class at the moment India most needs access to effective obesity treatment.
Timeline: How India Got Here
The Human Story Behind the Headline Number
Priya, 34, is a software engineer in Bengaluru. She asked not to use her surname. She started on Rybelsus eight months ago after her diabetologist suggested it alongside metformin for blood sugar management. She lost 11 kilograms. She feels better. Her A1C is down. But she is acutely aware that the drug is doing work she knows she should also be doing through diet and exercise - and that she hasn't been able to sustain either discipline during a period of heavy work pressure.
"I know it is a crutch," she says. "But it is a crutch that works. When the generics come and price drops, I will probably stay on it longer. Right now, I ration myself because of the cost."
Rajan, 58, works as a security guard in Chennai. He has Type 2 diabetes and was told by his government clinic doctor that semaglutide would help him. The clinic did not stock it. The private pharmacy quoted him 11,000 rupees for Ozempic - roughly a third of his monthly salary. He is managing with metformin and dietary advice he half-follows. If generic semaglutide reaches 3,000 rupees, he says, he will find a way to pay for it.
These two people represent the actual scale of who the patent expiry reaches. Not the body-conscious professional who wants to drop 10 kilos before summer. The person managing a chronic disease that is genuinely shortening their life, who has been priced out of a treatment that might help.
India has more than 77 million people with Type 2 diabetes. For a large fraction of them, cheaper semaglutide is not a lifestyle choice. It is access to medicine that exists and works but has been priced out of their reach. That is the real headline buried under the Ozempic-craze narrative.
"The arrival of these drugs has changed the conversation - obesity is now being treated as a disease, not just a lifestyle issue." - Dr. Muffazal Lakdawala, bariatric surgeon, Mumbai, via BBC
What Happens Next
By summer, India will have more brands of semaglutide than most countries have pharmacies stocking it. The competitive price war will benefit patients who are well-informed, medically supervised, and stable in their access. For everyone else, the picture is messier.
India's government needs to move fast on three fronts. First, manufacturing oversight - the Central Drugs Standard Control Organisation needs the resources and the political will to inspect the wave of new production facilities. Second, prescribing controls - the current rules about prescription-only status for GLP-1 drugs need enforcement, not just advisory circulars. Third, education - the social media narrative around these drugs in India has outpaced the medical evidence, and correcting it requires a public health communication effort that India has not yet begun.
The HIV parallel has a dark side worth naming. Indian generic antiretrovirals transformed global HIV treatment. But they did so within a framework of international partnerships, procurement systems, and quality monitoring built over years. The semaglutide rush is happening in days. The infrastructure is not comparable.
Dr. Baxi, the Mumbai diabetologist who turned away the 23-year-old gym kid, sees both sides. "These are medicines that change outcomes for real patients with real disease," he says. "But they are going to be used on people who don't need them, at doses they weren't assessed for, without supervision, and the people who get hurt will probably be the ones least equipped to deal with the consequences."
Thursday, India crosses a threshold. The drug that became a cultural signifier of wealth and aspiration in New York and London becomes, at least in theory, accessible to the woman in the government clinic who cannot afford what her doctor prescribes.
Whether that access translates into benefit or catastrophe will depend on what happens in the 90 days after the patent expires - in the manufacturing plants in Hyderabad and Ahmedabad, in the enforcement offices in Delhi, and in the 23 million pharmacies and informal drug vendors scattered across a country that is, in every sense, too large to regulate from the top down.
The magic pill moment is also a stress test. India has passed versions of this test before. Whether it passes this one will matter not just for 180 million people at home, but for the hundreds of millions more in the developing world watching what India does next with a molecule that could genuinely extend their lives.
Get BLACKWIRE reports first.
Breaking news, investigations, and analysis - straight to your phone.
Join @blackwirenews on Telegram